 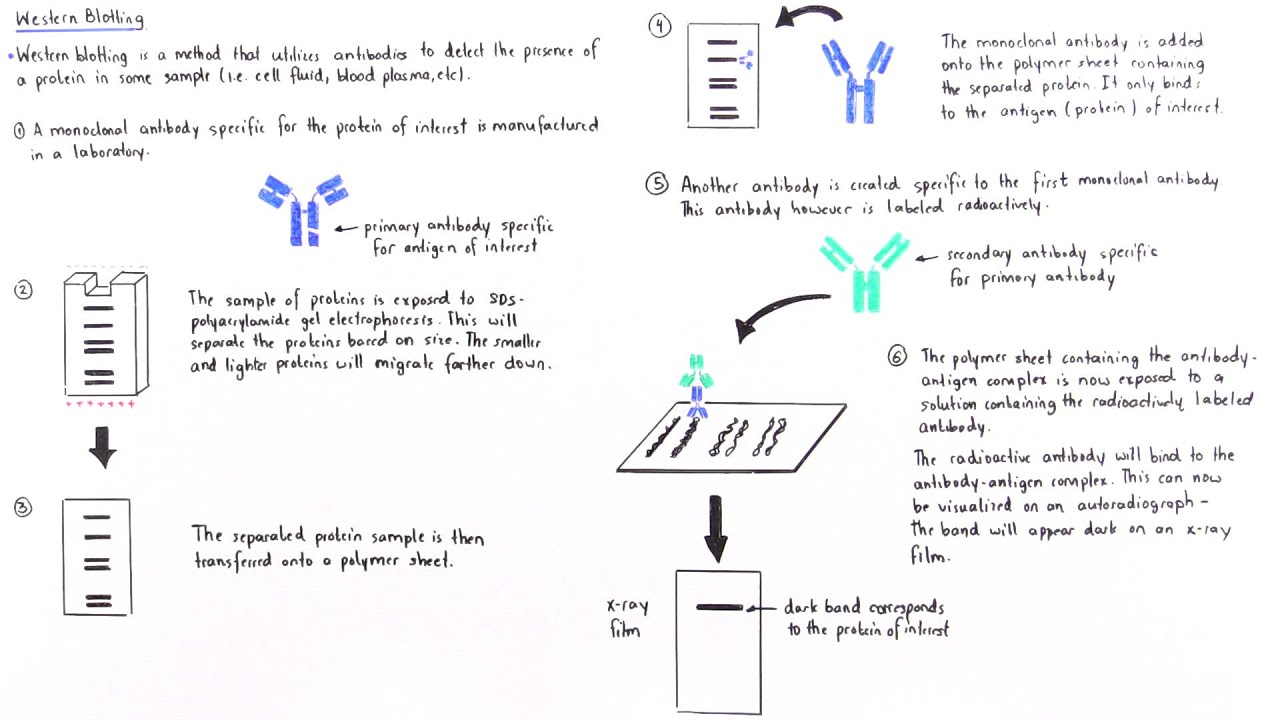 
Large overhangs may prevent a current from passing through the membrane in semi-dry transfers.Ĭhicken antibodies tend to bind PVDF, leading to high background. Make sure the paper and membrane are cut to the same size as the gel. Proteins on fingers will block efficient transfer and create dirty blots.Īfter sandwiching the gel and membrane between paper, air bubbles between the gel and membrane can be removed by rolling them out with a pipette, roller, a 15 mL tube, or by assembling the sandwich in a dish of transfer buffer to prevent formation of bubbles in the first place. Set the parameters and time.Īvoid touching the membrane with your fingers. For semi-dry transfer, the gel side should face the cathode plate (-), while the membrane side should face the anode plate (+). For wet transfer, the gel side of the cassette holder should face the cathode (-) while the membrane side should face the anode (+). Place the sandwich into a transfer cassette and perform semi-dry/wet transfer. Note: Bubbles between the gel and the membrane will inhibit the transfer of proteins to the membrane. Gently remove any air bubbles with pipette. Sequentially assemble the layers of the sandwich. Prepare the sandwich according to the illustration below. Once electrophoresis is complete, remove the gel from the electrophoresis apparatus and equilibrate it by soaking it in transfer buffer for 5-10 mins. Soak filter papers and sponges in the transfer buffer for 5-10 mins prior to assembly of the transfer sandwich. Handle the membrane carefully, ideally with rounded tweezers to avoid scratching or puncturing the surface. Prepare the PVDF membrane: wetting it in methanol for 30 seconds and then soaking it briefly in distilled water followed by 1X transfer buffer. Over-transferring can occur and thus caution must be taken, especially for small proteins. Tips: Transfer time and voltage may require optimization. Generally faster, better suited for larger proteins greater than 100 kDa. Recommended for smaller proteins, especially proteins smaller than 30 kDa. 
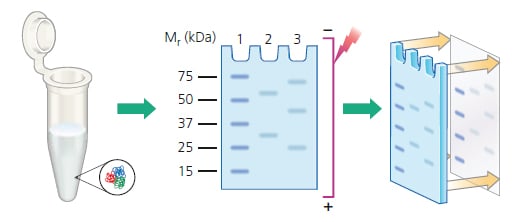
In contrast, large proteins can be slower to elute and may be retained within the gel, so overnight wet transfer is usually preferred. If there is a suspicion that the protein is transferring through the PVDF membrane, then a second membrane can be included behind the first to catch proteins that migrate through. PVDF with a smaller pore size can be used for small proteins and peptides. Smaller proteins will transfer out of the gel faster and may actually transfer through the PVDF membrane into the filter papers beyond. The size of the target protein should be considered when choosing transfer conditions. In a wet transfer, the gel/blotting paper/filter paper sandwich is placed into a cassette along with protective fiber pads. Electrophoretic transfer can be accomplished under wet or semi-dry conditions. The transfer buffer used for electroblotting is similar to gel running buffer with the addition of methanol which helps proteins bind to the blot. The gel and membrane are assembled into a sandwich along with several sheets of filter paper which protect the gel and membrane and help to ensure close contact between their surfaces. Taking PVDF membrane for example, the PVDF membrane is placed between the gel and the positive electrode so that the negatively charged proteins migrate from the gel onto the PVDF membrane. In general, PVDF is much better for low molecular weight (MW) proteins. Compared to NC membrane, PVDF membranes offer better protein retention, physical strength, and chemical compatibility. There are two common membrane types used for western blot (WB) analysis, polyvinylidene difluoride (PVDF) and nitrocellulose (NC). Transfer is performed by passing a current across the gel to the membrane. Due to the speed and efficiency of transfer, electroblotting is the method relied upon in most laboratories. Protein Transfer from Gel to Membrane in Western Blot Assayįollowing gel electrophoresis, the separated protein mixtures are transferred to a solid support for further analysis. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
